|
Psst. Hey kid, wanna learn about Decadent Literature, The King in Yellow, and the story collection that helped launch Lovecraft's own writing and weird fiction as a genre?
0 Comments
Well, it happened. Keeping for two years, six months, and a few hours in change give or take, and it finally happened to me. I got sick and the worst of it happened while I was Keeping a game. Oh, it's not like it hasn't ever happened before, at least not in the sense of some very close calls. I once had to run the final session of a Nyarlathotep campaign while dealing with the aftereffects of losing my voice from a cold, which ended up making my normally soprano-ranged voice much scratchier and huskier than normal... not at all a bad vocal look for my sleazy, captivating, and darkly sensually attractive (even the guys want him!) interpretation of the Black Pharaoh himself. In fact, I daresay it made the scenario's end better, if not for the fact I basically sucked down more than my fair share of bottled teas and juices to stay hydrated. On that note, caffeine is bad for your voice, don't do that thing before you GM anything - just sayin'. To be fair, I should have known - there was a nasty virus going around, some rhinovirus strain or another that had spontaneously mutated, and that nobody had any antibodies to. Being on a college campus almost 24/7, and my siblings also being so to a point, someone in my family of course caught it. Then I caught it. Then my labmates in my Nucleic Acids lab all caught it, including my poor professor who also teaches my OTHER CMB lab, which all then got it, too. And yet, none of my CoC group caught the little fucker, despite openly sharing and drinking from my root beer while the virus was starting to develop. I thought it only a little bit of a dry, sore throat at the time, caused by speaking for long periods. That does happen when one narrates, talks, screams, yells, jabbers, etc. across a table at people for hours on end, which is why having fluids on hand to hydrate thyself is always a good idea. I thought it was nothing and proceeded to ignore it. Oh good Lord, I couldn't have been more wrong. I was sick the entirety of Labor Day Weekend, not that long after my birthday. My throat was raw and felt like acid burns, my nose and sinuses were a runny, aching, stuffy mess, and I generally felt like shit for three days. The fourth day, I thought it was over. I thought I'd be okay and that the worst of it was leaving - my throat was better, my sinuses were clearing a little, the post-nasal drip had dried up. It had to be over, right? Ahahahaha, no. Nope. Not even fucking close. Instead, the little bastard decided my lungs would make a lovely place to raise a family of approximately billions of little virion particles, and it set up shop in my bronchioles and proceeded to start drowning me in my own fucking mucus. I literally couldn't breathe walking from my car to class and was tied to a fucking inhaler the entire rest of the week. I couldn't even walk up the damn stairs without hacking and wheezing, and of course, my nose was stuffed up to compound my inability to get any fucking oxygen. This lasted for another four fucking days, and only just this past week was I able to feel better. I was tied to cold medicine and a nasal spray in order to even function and get through my classes, on the very worst week of the semester for my Nucleic Acids lab (I was going in every single day, no fucking joke, outside of class to prep things and streak things and run gels and ARGH FUCK WHY IS THIS MY MAJOR AGAIN). It was hell, absolute hell, and only caffeine and sheer willpower got me through it. Well, and this lovely little chemical compound, of course: That little buddy right there is a wondrous compound by the name of (4bS,8aR,9S)-3-Methoxy-11-methyl-6,7,8,8a,9,10-hexahydro-5H-9,4b-(epiminoethano)phenanthrene, but its friends call it Dextromethorphan or "Dexy" for short. And now you all just realized where that band that did "Come On Eileen", Dexy's Midnight Runners, got their name from. You're welcome. Dexy here is your friendly neighborhood cough suppressant, it's found in most cough medicines you get over the counter and it's also found in many Dayquil/Nyquil type products. I'd wager to bet this guy was your friend at some point in time while you were ill, but you never even bothered to learn its name, you selfish motherfucker. Of course, Dexy isn't all seriousness and sickness. No, it has a... wild side. A party side, you might say. That is, it's used as a recreational drug by some folks. There's a popular way of consuming this drug that involves cough syrup and Sprite or vodka, known to some as "Jungle Juice", "Sizzurp", or "Purple Drank". Take too much of it, and you'll start to wonder where the fuck you are. You see, Dexy here is a semi-powerful disassociative drug, and acts as a bit of an analgesic - that means it makes you high as balls, unable to focus, and numb in the extremities. Those who do this on purpose call the phenomenon "Robo-tripping", supposedly because it makes you feel like a robot - cut off from the world, experiencing everything slower, and reacting without much emotion; in other words, you dissociate and things don't feel real anymore. You can actually experience anything from stimulant-like effects to mild hallucinations, depending how much you take, as well as nausea, vomiting, confusion, nervousness, and drowsiness or dizziness. These effects usually occur over the course of several hours with many "peaks" or "plateaus". In other words, that means you'll not just experience this bullshit once, but you might experience it several times over the course of one night. You could be fine one moment, and then the next you are out of it and unfocused. Some people would call this "awesome". I however call this a nuisance and vaguely frightening, because I am unabashedly straight edge and every time I've had experience with any sort of altered state it has never been pleasant for me, at all, ever. So, let me tell you about how I, doped up on way too much Dexy, Robo-tripped my way through a CoC session last week and why I don't have any material for reviewing "The Spawn" as a result. Now, I gave my players fair warning - I could feel that I wasn't right in the head, and that both my sickness and the medicine were doing this to me. I knew I was tripping, because I knew I took way too many cold meds (the Dayquil type ones) and hadn't let them wear off between before I took another dose of two pills. Granted, I wasn't coughing and hacking, and I wasn't knocked out moaning in suffering from how bad I felt, but I still wasn't there. I'm honestly surprised I got the Investigators on the goddamned train, off to Coppertown in New Mexico, and off the train. I didn't even characterize the town. I half-assed the map and only changed it from one long street when my brother said something to the effect of, "Wow, nice town, GM". The map wasn't the greatest either, but to my addled mind it looked just fine. Until someone asked where in the fuck the police station was, and I turned to look. The only response I had to forgetting to add it was, "Oh." Nothing else, just "Oh." And then I sloppily wrote "P. Station" on the poorly made square, added a star, and called it a day on the map. I'm surprised nobody commented on the Pee Station, or called it a Playstation. Maybe they realized I was just that out of it. To be honest, I really don't remember what the fuck everyone did in Coppertown, except for about two things. I remember them meeting Jose Verde, my Chicano take on Jose Green in the scenario proper, and I remember them talking to Dr. Tyler Freeborn briefly. They also visited some of the miner's outposts in a very, VERY roundabout way, no doubt not helped by my not being able to remember where in the fuck they could drive, where the railroad tracks for the town were, and why they couldn't take their beat-up struggle buggy of a van up the mountains... or just walk. It seriously took me five minutes to give a decent, coherant explanation as to why they, no, could not walk several miles up mountains in thick Ponderosa pine wilderness they had no familiarity with to get to the mines. And all this while feeling like that kid from David After Dentist. Sadly, it was indeed real life, and I had to continue. The players chose to follow up on Jose's lead and go explore the graves of the freshly interred Broad Vein miners, killed in a supposed collapse. They all dug up all the graves and saw the weird circular burn marks on the bodies, and of course were nearly caught doing it. Thankfully, I bungled the absolute fuck out of the rolls for the guards near the graveyard, and nobody was caught. I was even nicer than normal to my one player who just, for the life of him, could not Stealth to save his skin. I probably should have let him suffer, but eh, I could barely tell whether or not the dice were candies at this point (I have very brightly colored, pretty dice, and the ones I was currently using were bright yellow like lemon drops). Not to mention that I was laughing at like every dumb joke way too hard for way too fucking long because OMFG that joke about the holes being for necrophiles was just too fucking funny you guys comedy platinum right here hashtag CoC: RotOO squad 2016. The game continued like this for a good. Five. Hours. I have no idea how I even made it through and no idea how it turned out because I have literally almost no memory of that whole night (voluntarily blocking the horror from my conscious mind?), but I know it was basically a filler/establishment session. There was nothing I could do to make it not be that way, because I couldn't even tell whether or not my hand was real, let alone what the fuck a Copper Lady was supposed to be. You might as well have asked me where I was, and I would have answered, "I don't fucking know, Lost Carcosa, because I keep hacking up pieces of it from my lungs every five seconds?" And I would have, in all seriousness, believed that I was standing in Carcosa, and you need to take your mask off right now, because your face looks not real at all, and neither does my face, and oh my fuck I am so sick and I don't understand why this is happening to me dear god help me. I have no idea why my friends let me continue Keeping that night. They really fucking shouldn't have, because immediately, after driving my friend home (which, OMFG, how?), proceeded to crash on my bed and sleep like a dead woman for the next three days. Moral of the story, kids? Dexy and Keeping do not mix. Just... just cancel the session if you're that sick. Trust me, everyone will understand. Don't powerhouse through it. Just don't do it. Go to fucking bed, set down the dice, and call it a day. It will not end well otherwise.  Guys, I am a serious nerd, and if you've read this blog for any length of time you're very likely aware of this. You should also be aware, my dear and dedicated Muselings (that is the technical term for those Internet users who read my humble little blog, a term invented as of five seconds ago), that two of my great loves on this site are Chemistry and Pokemon. Now, I've discussed Pokemon on this blog before, although that discussion concerned anti-Pokemon and anti-Pokefan sentiments regarding such biased and ignorant reasoning as "there was a symbol that looks sort of like a swastika on the card Koga's Ninja Trick, therefore Pokemon supports Nazi ideals" and "Pokemon are revived when they have fainted, so they must be demons that can be brought back from the dead! Clearly Pokemon supports Satanism - never mind that said belief was only the opinion of a few extremist Evangelical pastors and was never actually supported by any sort of Biblical or Scientific fact". I've made it pretty clear, I think, that I love Pokemon despite its flaws (WTF, you fight little creatures you capture until either it or its opponent faint, and then you get money for it - I'm sorry, that's an awful lot like dog-fighting, and the only reason I find this premise acceptable is because the games and the anime/manga both make it clear that a trainer should raise their Pokemon with love and care outside of battle), and would be one of the first people to defend it should someone object. Because I'm a dedicated Pokefan, one site that's a constant companion to me is Bulbapedia, a Wiki for everything Pokemon. Recently, I had restarted Pokemon Crystal version and was looking up how best to carefully raise a Level 20 Eevee so it can evolve into Umbreon by quickly raising its Happiness without leveling it up too much and losing some of the precious Dark-Type moves Umbreon can learn. Happiness is basically a measure of how strongly your Pokemon has bonded with you, and some Pokemon only evolve into their next form if they are very happy with you as a trainer. Umbreon and its Psychic-type sibling Espeon are two of the many Pokemon obtained by raising a Pokemon's happiness (in this case Eevee) to or close to its maximum level (Evolution of happy Pokemon begins at a Happiness stat of 220 or so) and then levelling it up (Eevee should take a level at night for Umbreon, and during the day for Espeon). I specifically wanted Umbreon because Dark-Types have some strong moves and high defense, making them absolute tanks in battle. Plus, Umbreon is a badass, seriously, just look at it. It has no fucks to give. Ever. But anyway, a Pokemon's Happiness can be decreased in several ways, such as by letting the Pokemon faint too much, as well as increased, including by just walking around with them in your party. Another way to increase Happiness is by feeding the Pokemon Vitamins, which are battle stat-enhancing items that can be used to permanantly increase things like Attack, Defense, Speed, and Hit Points (HP). The advantage to this type of Happiness-leveling is that not only does the Pokemon like you more, it has better stats in battle, making it a win-win scenario for any trainer looking to obtain a Pokemon that evolves through Happiness. The disadvantage is that all the Vitamins are quite expensive, costing around 9800 Poke-dollars per item, but the benefits are well worth it. Of course, real vitamins don't work this instantly or permanently, but Pokemon takes place in a different universe anyway. And now you know how I came to find the page for Vitamins on Bulbapedia, and thus how I came to write this article. You see, I discovered an interesting little factoid concerning the Vitamins - in Japan, many of them have the names of real life chemical compounds! I then became intrigued and began to wonder just how many of those compounds really do in real life what the Vitamins in Pokemon do for your Pokemon. So hang onto your Pokeballs and make sure your Jigglypuff is wearing the proper lab gear, because science is about to get weird. As of the Generation V games (Pokemon Black and White versions along with the upcoming Pokemon Black 2 and White 2 versions), there are nine different Vitamins you can feed a Pokemon. Five of them increase battle stats, one increases HP, two increase Power Points (PP) for various moves, and one increases level. It is the five Battle stat-enhancers - Iron, Calcium, Protein, Zinc, and Carbos - that have real life supplement names. Specifically, in Japan:
Taurine, or 2-aminoethanesulfonic acid, is an organic acid found in most animal tissue and that many animals need to function. For example, cats need Taurine to keep their eyesight sharp, and if they don't get enough of it they can go blind. Taurine is a component of bile, and it's essential for your eyes, heart, muscles, and brain to fucntion properly. It was first obtained from Ox bile in 1837, and this is where it gets its name (from "taurus", the Greek word for "bull"). It occurs naturally in seafood and meat, although even if you're a carnivore, you're likely only getting around 200 mg per day of the stuff. In terms of Pokemon, which are based upon real animals, Taurine might very well be essential stuff for them, particularly for Bird-Type (like Pidgey), cat-like (such as Meowth), or carnivorous (like Houndoom) Pokemon. It may serve the same function in Pokemon that it does for animals - eye, cardiovascular, and muscle health - and some Pokemon may very well need it to develop properly. There is evidence that insect-eating songbirds, such as sparrows and robins, need it to develop right, and these birds often seek spiders (which are rich in Taurine) to feed their young. And of course, every mammalian creature needs it for their eyes and muscles. Therefore, it makes sense that Taurine would be present as a Pokemon Vitamin - it would help the Pokemon see in battle better, hit harder due to its stronger muscles, and have better endurance because of the increased cardiovascular health. As for Bromhexine, it is used for respiratory disorders, specifically for the treatment of mucus, and it has antioxidant properties. I can't see how this would benefit a Pokemon's Defensive abilities, but the antioxidant properties may help increase the Pokemon's resistance to diseases caused by free radicals, which would indeed help it be able to take more hits and therefore have a higher defense. Additionally, a Pokemon's body might use the molecule differently from a human being's, making it more beneficial to a Pokemon than a human. I don't know how Bromhexine is used in Japan, but it may be that the programmers of the Pokemon games exaggerated its protective antioxidant effects to legitimize its Defense-increasing properties, and this is probably why the Vitamin was renamed "Iron" in the English versions of the games. Naming the Vitamin Iron makes more sense anyway - iron is vital for the production of the oxygen-carrying hemoglobin, as well as for many cellular and immune system functions. If there is not enough iron in the bloodstream, a person (or Pokemon) can endure poor immune function, fatigue, weakness, and dizziness amongst other symptoms. It therefore makes sense that, by feeding a Pokemon Iron, you are actually raising its resistance not only to disease, but attacks in battle as well. Now to look at the Special Stat-raising items Zinc and Calcium. Zinc in real life is necessary for quick wound-healing and is beneficial for the immune system, which explains why it was chosen for the name of a Special Defense-raising item. Some also believe it possesses antioxidant properties, which further cements it as beneficial to a Pokemon's defense. This Vitamin in Japan is called Chitosan, which is a polysaccharide (a large sugar) obtained from the shells of crustacians such as shrimp and crabs. Chitosan has been shown by the US Marine Corps to quickly stop bleeding - in fact, when tested on pigs, it allowed a wounded animal to survive otherwise lethal bleeding 100% of the time, since it reduces blood loss by quickly clotting the blood. In fact, Chitosan was recently approved in the United States and Europe as an additive to hospital bandages as well as to products used on the battleground by both the US and UK military - in fact, this compound was used during the war in Iraq and Afghanistan by US and UK soldiers to stop bleeding on the field. Additionally, Chitosan has antibacterial properties and is hypoallergenic despite being derived from shellfish. Someone at Nintendo certainly did their research, because not only do both Chitosan and Zinc support wound-healing and quick blood-clotting, they are both excellent candidates for defense-raising items, and I don't think I need to spell out just how they could benefit a Pokemon that ingested them. In fact, maybe Zinc/Chitosan should have been the item that raises Defense, and Iron/Bromhexine the one that raises Special Defense! Calcium, on the other hand, supports not just strong bones but also cellular processes and proper nervous system function (it is necessary to transport signals between nerve endings to the brain and back). Therefore, Calcium could increase Attacking power by giving skeletal and nervous system support to a Pokemon - and a Pokemon with strong bones and a healthy nervous system can fight better and hit harder by virtue of having quicker responses and a stronger skeleton to support their muscles. In Japan, this Vitamin is named Lysozyme instead. Lysozymes, which are not one specific chemical compound but a group of them, are named for their lytic, or cell-destroying, properties ("lysozyme" literally means "an enzyme that breaks things apart"), and they are an important chemical produced by healthy immune cells. Lysozymes work by cutting apart bonds between molecules in a bacterial cell's cell wall, which causes the bacterium to lose structural integrity and literally fall apart. This in turn kills it, and the benefits of killing invading bacteria are fairly obvious. Some areas of the body produce a lot more of these compounds than others - for example, the eye produces excessive amounts of lysozymes to kill bacteria that get onto the conjunctiva (the white part of the eye), and when these enzymes fail the result is a bacterial infection called conjunctivitis (commonly called pink eye). Therefore, it seems obvious why you'd want to feed a Pokemon lysozymes - it boosts their immune system, and when you have Pokemon that can poison you or attack you with spores, it makes good sense to ensure a Pokemon isn't too terribly damaged by these attacks. Plus, by keeping bacteria out of your Pokemon, you're ensuring it doesn't become sick and therefore have a harder time battling. Once again, someone at Nintendo clearly did their Chemistry homework. Finally, we'll look at Carbos, the Speed-increasing Vitamin. The word "Carbos", as if I even really have to say it, is a shortening of the word "Carbohydrates", which provide energy and are the most abundant source of fuel in most animals' diets. In fact, very complex carbohydrates can make you feel very full for a long time, such as those present in wheat breads and oatmeal. Smaller carbs, such as those found in white breads and sugary foods, burn quickly, providing a very fast burst of energy which wears off too soon. Both types of carbs are processed by the body for energy, which is why many athletes eat meals high in carbs before a big game. Therefore, it seems pretty obvious why Carbos would promote a Pokemon's speed - if a Pokemon eats a lot of carbohydrates, they'll have more energy in battle and be able to move and react quicker. After a while, their body (like with humans) would get used to burning a lot of energy, thus they would need more carbohydrates to stay alert and active because Pokemon lead such a high-activity lifestyle. Of course, some Pokemon, like the speedy Zubat, might burn carbs more efficiently than others, like Slowpoke. Therefore, a faster Pokemon would need more carbs in their diet than a slower one, because a faster Pokemon would burn those carbs much faster. In Japan, the Vitamin Carbos is called Indometacin. Indometacin is an NSAID (Non-Steroidal Anti-Inflammatory Drug) that is used to treat pain, stiffness, and swelling as well as Fever. It can poison in very high doses, so extreme care must be used to ensure the proper dose is given. Because of its potency, it is only given in cases of severe or chronic pain, such as with rheumatoid arthritis or tendinitis. Pain, stiffness, and swelling are all symptoms of injury that a Pokemon could endure in battle, and severe pain and stiffness could indeed reduce Speed, so it seems clear why Indometacin would be a good supplement for a Pokemon - by increasing pain resistance, such as that caused by constant battles, a Pokemon can easily become speedier - just ask any arthritis-sufferer how much more quickly they could move were they not in constant pain. In fact it was probably this pain-killing property of Indometacin that caused the English version of the games to change the Vitamin's name to Carbos instead, which still preserved the Speed-enhancing aspect without the implication that Pokemon can develop chronic pain and stiffness from heavy battle. Even if the idea of it happening is more realistic, Pokemon arthritis is a pretty heavy-handed concept for a kid to grasp. So, that explains those five Vitamins, but how about the HP, PP, and level-raising Vitamins? Here's where we get into speculation about what compounds these Vitamins actually are, since nobody at Nintendo seems to have said anything about them. I'll assume that they would be based on real chemical compounds, going off of the assumption that the Pokeverse's chemistry is essentially the same as ours. I'll start with HP Up first. HP, being a measure of the amount of hits a Pokemon can take before fainting, is essentially a measure of that Pokemon's endurance - the higher the HP, the more damage a Pokemon can endure before being unable to take any more and collapsing in exhaustion and pain. Therefore, HP Up probably contains a mixture of, or a compound that, can increase endurance. Caffeine, one of the most obvious choices, has been proven to help because it aids muscle contraction and boosts aerobic endurance - a cup of coffee before exercise has been shown to increase how long and how far an athlete can go. Creatine, which is naturally found in fish and lean meats, is another good candidate, since it improves both anaerobic and aerobic endurance. Beta Alanine, another endurance-enhancer, is found in meats and can help boost muscle performance, particularly in high-intensity activities. Of those three, it seems most likely that HP Up is probably Creatine, but could include some Beta Alanine in it as well. As for the PP-increasing Vitamins, it seems likely that those contain Caffeine, since PP is just a measure of how much energy a Pokemon has for a given move. After all, what better to increase your energy than Caffeine? If this is the case, then both of the PP enhancers have different amounts of Caffiene per pill, since one only increases a movie's PP while the other maxes it out. So using a PP Up is probably like giving your Pikachu a cup of coffee, while using a PP Max is like giving him a cup of espresso. Might not want to give him too much though - the last thing we need is an overly hyper Electric-type Pokemon getting so excited that he starts Volt Tackling around the living room, zapping every living person within a five-foot radius. Finally, that prized of all items, the Rare Candy. Because this item raises a Pokemon's level, it could be one of several things, or a mixture. We do know that Rare Candies come from Berry Juice, which comes from Berries being held by a long time by the Pokemon Shuckle. Obviously, there is something in the Rare Candy, then, that is also present in the health-giving item Berry (this was before Generation III introduced the different types of berries; according to Bulbapedia, Shuckles generally tend to hold Oran Berries after Gen III, which restores 10 HP if a Pokemon eats it). A possible explanation for why Oran Berry Juice, which then crystalizes and becomes a Rare Candy, is that Oran Berries are high in vitamin C like their citrus counterparts in the real world. Vitamin C is supposed to strengthen immune system responses. Some citrus fruits, such as mandarin oranges, are also higher in certain sugars than others, and since the Oran Berry is likely based on this Asian-native fruit, it is likely that the reason Pokemon gain a level and thus get stronger from eating a Rare Candy is due to the extra sugar and Vitamin C in the Oran Berry, or at least is based on that idea. This also explains why it raises a Pokemon's Happiness - not only is it becoming stronger and growing up, it's also eating something that tastes sweet. And as we all know, it's impossible not to like someone who gives you candy, provided they aren't some skeevy fellow in a Pedo-van. However, there are alternative explanations for the Rare Candy's abilities. On one hand, a Rare Candy could contain a mixture of all the other Vitamins, effectively making it a Pokemon multi-vitamin which could promote the Pokemon's quicker growth. However, this seems unlikely because Rare Candies only increase a Pokemon's level, not its battle stats. On the other, much more likely hand, a Rare Candy could contain Pokemon growth hormones - basically, Pokemon steroids. The only thing that would make giving a Pokemon an extra level in this manner legal, then, would be if it were such a small amount of the hormone that it only raised the level by one. If this weren't the case, then it would mean a Pokemon could become ridiculously strong, ridiculously fast, with the added downside of being ultimately weaker than a normally leveled Pokemon due to the effects of chronic steroid use. This is supported by the fact that only the Pokemon's level is increased, and by the fact that a Rare Candy can revive a fainted Pokemon due to the increased HP it takes. In fact, there may even be an illegal underground Pokemon steroid trade, which certainly explains why big cities like Celadon and Goldenrod need their own police departments. I smell a Pokemon urban legend in the making, guys... Quickly, reader! Find someone who hails from 4Chan or SomethingAwful and get them to make a Copypasta out of this! It must be done FOAR GREAT JUSTICE. Well, that's it for Weird Science today. Make sure you level up your Pokemon before that next Gym Leader battle. As for me, I've got an Eevee to entertain, so I'll catch you guys later (but not in a Pokeball though seriously why would you even think that?). 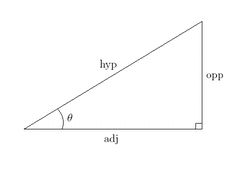 Hi there, and welcome to yet another installation of Weird Science, a series of blog entries here on Musings where I take a look at the fictional and apply scientific and mathematical fact to it. Today, I'm headed into the forest lovely, dark, and deep to tackle the trigonometry of the Slenderman. Care to join me, reader? Yes? Well, alright - just make sure you stay close! Stragglers never fare well in these woods, especially when it's night time and they're all alone... Now, as my more loyal Muselings will recall, I love discussing mathematics and science. Now, usually that pertains to statistics and chemistry, but what some of you might not know is that I'm pretty good at trigonometry, too. I can judge angles by looking at them. I can solve for the sine of theta and the number of radians in any given degree with the best of them, and I fully believe that just about anything measurable can be solved with a little trig. So today, I'm going to apply that trig knowledge to a favorite horror fandom of mine, and specifically a scene in my short Slenderfic The Hunted. For those who haven't read it yet, this blog entry is spoiler-free, but does discuss the story's beginning. If you'd like to read The Hunted for context before you read this blog entry, you can do so by clicking right here, but no context is needed to understand what I'm doing here. Let's start with the scene from the story in question - my protagonist, Jeremy, up in his hunting blind which is eight feet off the ground, looks out of it to see Slendy standing about 30 feet away from him. He is able to look the being in the eyes - or he would be able to, anyway, if Slendy had any eyes to look into. Jer, confused and more than a bit startled at the apparent height of the being, looks down at the ground at an angle of 17° from horizontal to double-check what he's seeing. The question is, can he estimate how tall Slendy really is by doing this? Before we answer that question, let me give you guys a little right triangle trig review, since we'll basically be working with right triangles anyway. The right triangle has three parts - an adjacent side (sometimes called b), an opposite side (sometimes called a), and a hypotenuse (sometimes called c). You can find one side if you have the other two by using the Pythagorean theorem, which states that a^2 + b^2 = c^2. That funky-looking little O with a line through it is the Greek letter Theta, and it denotes an angle that isn't a right angle. Hopefully, you remember that the square in the corner means a right angle (which measures 90°;), while the arc next to theta means any other angle. You can find the a missing side if you have only an angle and a side by taking the sine (opposite/hypotenuse), cosine (adjacent/hypotenuse), or tangent (opposite/adjacent) of the angle in question, depending on what side it is you want to find. You can find a missing angle by taking 180°, and subtracting the other two angles you do have from it. Now onto the question at hand. Could Jeremy accurately measure Slendy's height with trigonometry based on what he observes around him? The answer is yes, of course - but how? Well, let's look at what we know. First off, we know that Slenderman is going to be be making a right angle with both the ground and its line of sight to Jeremy, since it's standing 30 feet from Jeremy and is also standing straight up and down, as Slendermen are wont to do. Jeremy can also look down at an angle of 17° to see its feet. This makes a triangle with an unknown opposite side (Slendy's height), an adjacent side 30 ft long, and an unknown hypotenuse that makes an angle of 17° from Slendy's feet to Jeremy's position 30 feet away. We'll call this triangle the SlenderTriangle. Note that the SlenderTriangle is missing two sides, but we can fix that later. As for Jeremy, his story's a bit different. We know that Jeremy's hunting blind makes a right angle with the ground, since the tree grows straight up, and we know he can look down at the ground at an angle of 17° when he's looking at something 30 feet away from him - in this case, Slendy's feet, or more likely, the foliage and trees obscuring its feet, since this is in a forest. This creates a triangle with an adjacent side of 30 feet and an opposite side of 8 feet, with an unknown hypotenuse between Jer and the ground that forms an unknown angle between Jeremy's position and the ground 30 feet away. We'll find this angle later - we can't definitively give the angle as 17°, because in the story Jer only says he can look Slendy in the non-face, not that Slendy is any specific height. That means the SlenderTriangle may not be a right one, depending on Slendy's actual height, but we'll assume it is one. As for Jer's Triangle, as we'll call it, we do know it's a right triangle and thus can apply right triangle trig rules to it. Notice that both our triangles need the same hypotenuse. Since Jer's Triangle has two sides, we can use the Pythagorean Theorem to get the hypotenuse. Because the SlenderTriangle's not completely right, we can't do this trick with it. So, setting up the Theorem for Jer's Triangle, we get 30^2+8^2=c^2. After that it's a matter of simple number-crunching to get our hypotenuse, which turns out to be 31.04 feet. We now have all three sides of Jer's Triangle and two sides of the SlenderTriangle. We can now estimate how tall Slendy would be, both with the information we found and the information given. Now, according to the very, very loose canon surrounding Slenderman and its Mythos, the entity in question can be anywhere from 6 to 12 feet in height at a given time, and can adjust its height at will. If we take a mean height of this range, we get 9 feet as the average height for our faceless friend, which is pretty accurate to how most people tend to depict it. So we're hopefully aiming for a height of 9 feet for the SlenderTriangle's opposite side, but a bit taller or shorter would also work, although hopefully not below 8 feet (too short for the blind's height) or above 10 feet (too tall for the blind's height). Given that range to aim for, let's look at Slendy's height. Since we have an angle and two sides, we could use the Pythagorean Theorem to solve for the opposite side. We could also use the Law of Sines if we assume the triangle's not a right triangle, but that's a bit too convoluted. So, assuming the SlenderTriangle is a right one, we can simply adjust the equation to find sine a bit and get our answer. Currently, if we stuck all the numbers in, we'd get the following: sin(17°) = opposite/31.04. So to solve for the adjacent side, all we have to do is move our hypotenuse value over. That gives us an equation that looks like 31.04 * sin(17°) = opposite, and if we solve this equation, we sure enough get a value of 9.08 feet for the opposite side. That means Slendy is, in this scenario, standing just a bit over its average height of 9 feet. An intimidating height for it to stand at to be sure, and definitely reason for poor Jeremy to have pause when he looks out of his blind and sees it standing there! This can also work the other way. If we wanted to give Slendy a mean height of 9 feet and measure the distance from it to Jeremy, we could take the Pythagorean Theorem and solve for the adjacent side of the SlenderTriangle, or we could multiply our hypotenuse by the cosine of our 17° angle. Either way, we'd get the same result - a 29.7-foot distance between Tall and Scary and our hapless hunter, Jeremy. This value is off by slightly less than a full foot, but it's still close enough to the 30-foot distance of Jer's Triangle to work for the proportions given. This is still assuming that the SlenderTriangle is a right one; if not and the adjacent side really is 30 feet, we'd need to use the Law of Sines to solve this - right triangle trig wouldn't work. Now there's just one more question to solve, since I promised I would - what is the exact angle that occurs between Slendy's feet and the hypotenuse, since Jer could also look down at them to help him calculate his suit-wearing stalker's height? To solve this, we actually need to use the tangent formula, which is found, as you recall, by dividing the opposite side by the adjacent side. Actually, we could use any right triangle trig formula we wanted to find this angle, but using the tangent is easier since we could have done that calculation before we got the hypotenuse anyway. So, following that calculation, tan θ = 8/30, but we need the angle, not the actual tangent of said angle. That's where the inverse of tangent comes in, which here means that arctan(opposite/adjacent) = θ. Therefore, if we plug in our numbers and solve, we find that θ = 15°. Well, actually, it more accurately equals 14.9°, but the value's close enough that you could still call it 15° and be safe. However, if you were to use that angle in a calculation here, you'd probably want to play it safe and go with the more accurate value. That's it for this installment of Weird Science. Hope you had a fun, safe trip through the forest, and - hey, did we lose a few back there? Oh... guess not. I can see them up in those trees over there. They look kind of dead, though, judging by the branches sticking through their torsos... what do you say we get out of here and leave well enough alone? I'd hate to lose any more valued readers... *looks up at the title*
... Really? That's the title I'm gonna go with here? Of all the titles for a Weird Science entry that I could think of, the first one that springs to mind, the first one that I choose, is "The Krailie Conspiracy"? Wow. I mean really, just. Wow. That title sounds like some sort of arcane, cryptic fan theory about Marble Hornets, or possibly even the title of a Marble Hornets fanfic. Alternatively, it sounds like someone mixed The Bourne Trilogy with Marble Hornets, and that, unlike half of the forest epileptic trees that grow wild in this fandom, not only kind of makes sense but is far too fucking awesome to exist on this sinful earth. Anyway, the reason I came here today is because I want to talk about something... off I noticed in a recent Marble Hornets entry. Specifically, Entry #69, wherein Tim and Jay go looking for something back where everything started, and find a bunch of burnt tapes. Seems an average enough entry from MH, right? Definitely sets up some important plot points with those tapes, too, but this isn't about that. No, this is about what seems to be an oversight on the part of the MH crew. You see, as much as I love Troy and co. and find them all to be pretty nice guys, they aren't trained to write scientific fact into their series about a tall, faceless creep. Granted, when you're using Slen- Sorry, I mean The Operator, you're not gonna focus on scientific fact, you're going to focus on telling a good story, and these guys are great filmers and storytellers, but not that great at science. What I'm saying is, they might not have done their research 100% with this one, and it has to do with how those burned tapes appeared in this entry. First, let me explain a little bit about recording stuff on tape for those younger Hornets in the audience that weren't born in the VHS era. Before DVDs became a thing, there were all sorts of formats for films, home movies, and TV show episodes. We tried just about everything to get our movies in a home video format, including giant laser-read disks the size of a damn tire rim that you had to flip over halfway and that didn't even hold as much data as a CD-ROM did. The most ubiquitous form of home video format by the time I was born in 1991 was the VHS tape. The way a VHS tape worked was through a magnetized plastic tape that held video data. Don't ask me how in the Hell it worked, because honestly I don't know, I was maybe 7 at the time. And when you were done watching the video, you had to rewind the tape back to the other side so you could play the tape again. Hence the old video store saying, "Be Kind, Please Rewind", because some assholes would rent movies, not rewind them, and then bring them back so the next person would have to rewind the tape to watch the film. It was annoying. Now, thing is, we used tapes like these in video cameras too - yes, just like Alex in Marble Hornets, we too had those little cassette tapes we recorded shit on, you stuck them in the side of your video camera in a flip-open thing, and you hit record, and it would record it on the tape. Then you could either replay it in the camera (if yours was a fancy flip-screen thing), or you could put it in a special VHS player sized cassette and watch it on your TV. If that's not enough, even longer ago we put actual VHS-sized cassettes in these big ol' cameras you had to hold on a tripod or on your shoulder, and... yeah. Technological advances really make things a lot easier, don't they? Now, here's where I have a bit of a problem with Entry #69 and its burned firepit tapes. The way the tapes burned should not have been possible, given the levels of heat even a small fire can get up to and the way the plastics in a VHS tape tend to melt. Let me explain this to you guys, because I know most of you out there probably don't give half a thought to plastics. First thing you should know is this: Plastics are all polymers, from the Latin words "poly", meaning many, and "mer", meaning unit. Many plastics we use in everyday life form chains of these little mer units, something like this: mer-mer-mer-mer-mer-mer, where each "mer" is one single repeating unit, and each dash is a covalent bond. depending on how organized or loose the chains are in the plastic, we can call a plastic amorphous or crystalline; in general, the more crystalline a plastic is, the more brittle it is, and the more amorphous it is, the more it tends to flow in weird ways, like chewing gum. Yes, chewing gum's a plastic, in fact, it's polyethylene in one of its most amorphous forms. Now you know why I don't chew bubble gum. These chains can also sometimes crosslink with other chains to strengthen the plastic and form nearly unbreakably strong bonds (which in fact, is how epoxy glues work), but I won't go into that because it's not important here. Look it up if you're intrigued. Now, VHS tapes, like the ones above and just like the ones Alex Krailie used in filming his disastrous student film project, are made of a couple different types of plastics. The casing is made out of polypropylene (a very tough and ubiquitous plastic), the reels are made from polyvinyl chloride (PVC, used in piping), I'd hazard a guess that the clear windows are polystyrene (a brittle plastic unless foamed) due to experience with how brittle they are, and the magnetized tape is a polyethylene (the same stuff found in garbage bags) ribbon coated with things called pthalates (which, due to their alignment on the polyethylene chain, can hold a magnetic charge, and therefore can be used to record data). That last type of plastic product is commonly called mylar. Now, judging by the kind of tapes Krailie used (the small ones you put in a handheld camera), I'd guess that the four plastics in most use here are the same as for the bigger VHS tapes - polyethylene (mylar), polypropylene, polystyrene, and PVC. This poses a problem when it comes to something called the glass transition point. In laymen's terms, the glass transition point (Tg) of a plastic is the point at which a melting plastic becomes brittle, like glass. This can also happen at super-low temperatures, because some plastics and other polymers have Tg values below the freezing point of water. Of course, melting point also differs for each plastic, and each plastic melts in a different way. For reference, here's the glass transition and melting points for the four types of plastic found in a VHS cassette tape:
Now for reference, the hottest your typical outdoor fire can get is about 1000 to 1,200 °C, since fires don't usually burn with white-hot flames in your typical campfire. The lowest temperature possible for a fire that has a visible flame is 525 °C, if the flame is just barely visible. It's possible Krailie used wood or leaves with a fuel or lighter-started fire to burn his tapes, judging by the small amount of damage that was inflicted on the tapes Jay finds (the ones that aren't completely melted). And therein lies the problem - the tapes Jay discovers in Entry #69 should be entirely useless to him, because all of the plastics used in the manufacture of tape cassettes melt at temperatures well below your average small campfire made from burning twigs and leaves. Even if Alex used coal for his fire, that's still a high enough temperature to decimate any plastic he threw into it almost instantly, even if the fire was burning with a relatively cold flame. So the question is, did Marble Hornets fail thermodynamics and polymer chemistry forever, or is there some kernel of possible truth to what allowed certain tapes to be saved? Well, the answer is yes, depending upon where the tapes were in relation to flame proximity. That is, how close where those tapes to the actual fire itself? Notice in Entry #69, Jay not only finds some tapes off to the side of the mass of destroyed burned tapes, but he also digs under them to find some tapes that were spared the full heat of the flames. This makes perfect sense - heat rises, including that of a flame, so it's feasible that any tapes far enough off to the side of the actual fire, or located beneath tapes that were actually burning in the fire, could be spared a good amount of the actual heat and therefore might not fully melt the casing. They might reach their respective glass transition points for each cassette casing, but it's likely that the actual tape cassette would survive. However, there's one problem here - the casing and tape reels inside could not possibly survive the actual heat, unless they were buried far enough under the actual burning tapes, or were really far off to the side. In fact, if you were to burn a VHS cassette, the plastic casing would melt first, then the tape reels, then the clear windows, and finally the mylar tape inside. You still couldn't salvage a fully burned tape even if you had the film; the heat would pretty much destroy it. So the answer is, yes, there's truth to what Jay finds in that firepit in the park - you really can have tapes that are spared the full wrath of a flame, if you get lucky and some are far enough away and the flame isn't very hot, but all this assumes that Krailie burned the tapes in such a way that they were all unsalvagable. However, as we see in Entry #69 above, this is very clearly not the case - a good handful of the tapes survived pretty much intact, and in fact the next two entries show footage from those tapes functioning perfectly fine and being almost totally undamaged (and they were probably enhanced by Jay, too, but you never know just how much actual work the guy had to do on them to make them function properly). It seems pretty obvious to me that Alex did not burn the tapes as well as he thought he did (hey, the guy clearly wasn't operating on all mental cylinders by this point in time), and with that, I now put on my Proxy Mask to start speculating as to what went on here. It seems pretty clear to me that Alex was in a rush to burn those tapes before someone (Jay? Hoody? Tim? Another former MH cast member?) found them, which leads me to four possible conclusions:
WARNING: This entry discusses the making of a dangerous corrosive compound, and explains the danger of corrosives. I am a trained professional and this article is for educational purposes only. PLEASE, DO NOT TRY THIS EXPERIMENT AT HOME.
------------------ So, there's a rumor going around again that, apparently if you put baking soda and hydrogen peroxide together with a little bit of Mountain Dew, it will make an impromptu glowstick. I have seen this claim go around millions of times, and as a Chemistry Tech and soon-to-be Chemist, this greatly disturbs me. Because you see, you can't trust everything you read online (said anyone with half a brain ever), and sometimes the stuff you do read online can be very, very dangerous. As someone with a chemistry background and plenty of (sometimes painful) experience with strong acids and bases, I notice things, things that could be dangerous, things that people without my background wouldn't know - because honestly, did you pay attention in Chemistry class in High School? Chances are, probably not. But at the very least you should know that acids and bases will burn your skin, and badly. As a result, I feel the need to point out that this pernicious rumor about the Mountain Dew glowstick is not only false (it was even debunked by Snopes!), but extremely dangerous. So put on your goggles and button up your lab coats, because science is about to get weird (in a safe way, of course). First of all, I will point out this - please, people, do not trust everything you see online. The above claim is 100% true, and shit like this gets people killed and severely injured. NEVER, EVER follow instructions to make something like this from online if you don’t know first thing about chemicals, you can seriously hurt yourself even if you know what you’re doing! Now for the science. Hydrogen peroxide, chemical formula H2O2, is a base/is alkaline and is the simplest of a group of compounds called peroxides. They’re called that because these compounds are very rich in oxygen (per- = saturated, lots of; oxide = oxygen is attached to it). These compounds are usually oxidizers, and hydrogen peroxide is a damn strong one if you don’t dilute it first. Oxidizing agents generally accept electrons, and the opposite, reducing agents, donate them. We also call them proton acceptors and proton donors, since they also make good bases and acids respectively. A good way to remember Oxidation and Reduction in these terms is OIL RIG - Oxidation Is Loss (of a proton); Reduction Is Gain (of a proton). This sounds like it’s harmless, but when a strong oxidizer reacts with anything, the reduction that occurs can break chemical bonds. This is the process many compounds with a double bond or C=O group undergo, such as the formation of esters. Oxygen itself is often found in many caustic, alkaline/basic chemicals, including many soaps, as the hydroxy group (-OH). This functional group is negatively charged in terms of electrons, meaning it readily gives a proton which makes it a good oxidizer and a good base. Meaning that, even though hydrogen peroxide is not an acid and baking soda is a harmless neutralizing agent, the hydrogen peroxide acts as one, and the result of the reaction is a very strong, very caustic base. This is the chemical equation for the reaction between hydrogen peroxide and baking soda: H2O2 + 2 CHNaO3 = 2 NaOH + CO2 +H2O H2O2 is hydrogen peroxide, and CHNaO3 is baking soda. CO2 is carbon dioxide, H2O as everyone knows is water… and NaOH is sodium hydroxide, more commonly known as lye or drain cleaner. Yes, this chemical reaction makes drain cleaner, and because sodium hydroxide is a very strong base and a lot of it is produced, the weak phosphoric acid in the little bit of Mountain Dew in the bottle will do jackall to help neutralize it. Even diluted sodium hydroxide is a hazard and can burn skin, and yes, in concentrated portions it will indeed eat through concrete. The reaction produces so much heat it can make the liquid in the bottle boiling hot in seconds, and agitating it by shaking the bottle only makes it worse because agitation speeds up the rate of a reaction, as well as making gaseous products come out of solution quicker. And since you have a sealed bottle with carbon dioxide already in it, if the reaction continues for a long time the carbon dioxide will build up, and the pressure can cause the bottle to explode. Leading to, you guessed it, NaOH-covered plastic shrapnel and boiling-hot NaOH going everywhere - on your skin, in your eyes, all over your clothes, all over the road and other people… in short, it is a disaster waiting to happen and nobody should ever do this under any circumstances, unless it is part of a chemistry lab or something and they have the proper protective equipment on. Even then, it should be done in a closed Fume Hood or with a Blast shield on to protect your face from base-coated shrapnel. Now you might say, “Hey, Sugary, wait a minute - they put hydrogen peroxide and baking soda in toothpaste, don’t they? And hydrogen peroxide is used to clean wounds all the time!” You are correct here, but take it from me - the hydrogen peroxide you have and the kind put into toothpaste is very dilute. That means it does not produce very much NaOH, but it does produce it. Most toothpastes nowadays do not have hydrogen peroxide in them, and if they do it is not in conjunction with baking soda, since this will erode the enamel on your teeth. If you’re still not convinced that this shit is a terribly bad, dangerous idea, here is what burns from hydrogen peroxide can look like: [x] [x] (GORE WARNING. DO NOT CLICK IF SQUEAMISH.) And here is what burns from NaOH can look like: [x] [x] [x] [x] (SAME WARNING AS THE ABOVE. DO NOT CLICK IF SQUEAMISH.) Bottom line? Do not fuck around with ANY chemicals, even household ones, if you do not know what you’re doing and what they will actually make. |
About The Blog
Welcome to Musings 2.0, my personal blog here on WordFlow! Here, you can find out what I'm doing now and where I'm going next, as well as get my thoughts on the Cthulhu Mythos, assorted sundry writing topics, and various scientific topics. Archives
January 2019
Categories
All
|

 RSS Feed
RSS Feed
